Last month, I wrote a post on the widespread use of statin drugs and the dangers associated with them. Statin associated myopathy and mitochondrial impairment are serious and wide-spread side effects of statin drugs.
As I stated previously, I strongly believe that people need to be actively involved in their own health care. This means embracing a healthy Mediterranean diet and lifestyle as well as cultivating a positive attitude. And it also means using appropriate botanical and nutritional support as needed.
A number of natural compounds have been shown to help mitigate the effects of statin associated myopathy and mitochondrial impairment. Those that I find most helpful include the following:
Rhaponticum (Leuzea carthamoides) and Anabolic Enhancing Ecdysteroids
Ecdysteroids are chemicals found in insects, certain water animals, and some plants. Because ecdysteroids have a similar structure to the male hormone testosterone, they are widely used by athletes as a dietary supplement for increasing strength and muscle mass and to improve athletic performance.
As far back as the 1970s, ecdysterone (an ecdysteroid) was reported to possess anabolic properties. When Russian scientists compared ecdysterone with the anabolic steroid dianabol, both were found to have a similar effect on muscle tissue.[1]
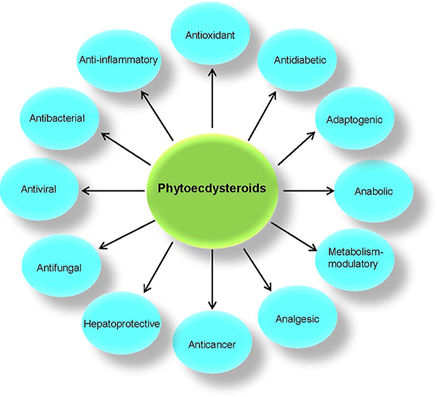
There is growing interest in the pharmaceutical and medical applications of ecdysone. Studies in vivo and in vitro demonstrate a wide range of beneficial effects in mammals, including anabolic, hypolipidemic, anti-diabetic, anti-inflammatory, and hepatoprotective.[2] In one study, researchers tested the effects of ecdysterone (5 milligrams per kilogram of bodyweight) on rats, and found that it triggered a faster rate of muscle growth than a similar dose of anabolic drugs.[3]
Ecdysteroids possess several favorable non-hormonal biological properties. The most interesting of these is the effect on degenerative diseases. Ecdysteroids exert adaptogenic action (protection of the organism against adverse stress factors) associated with anabolic, gastroprotective, and antioxidant properties.
A second group of favorable effects is the possibility of suppression of neurodegenerative processes and protection of the cardiovascular system (metabolic syndrome symptom suppression, antidiabetic activity, and protection of heart and blood vessels).[4] Studies show that ecdysteroids are cardioprotective and specifically aid in mitochondrial protection.[5]
The anti-inflammatory and antioxidant mechanisms of phytoecdysteroids also make them effective against various cancers. In addition, these compounds have demonstrated antimicrobial and hepatoprotective abilities.
Summary of Various Biological and Pharmacological Effects of Ecdysteroids
Rhaponticum (Leuzea carthamoides), rich in ecdysterone and other related anabolic compounds, is the most studied herb for its adaptogenic, anabolic and immunomodulatory effects.[7]
A 2009 review based on 117 literary sources discusses the current knowledge of traditional uses, chemistry, biological effects, and toxicity of this species. The extract preparations from the plant exhibit various additional biological effects, including antioxidant, immunomodulatory, anticancerogenic, antimicrobial, antiparasitic and insect repellent activities.[8]
Several different classes of compounds have been isolated from various parts of Rhaponticum. The main groups are steroids, particularly ecdysteroids, and phenolics (flavonoids and phenolic acids) along with polyacetylenes, sesquiterpene lactones, triterpenoid glycosides and terpenes (essential oil).
Resveratrol
Resveratrol is a stilbenoid, a type of natural phenol, and a phytoalexin produced by several plants in response to injury or when the plant is under attack by pathogens, such as bacteria or fungi. Sources of resveratrol in food include the skin of grapes, blueberries, raspberries, mulberries, and peanuts. Researchers have used resveratrol to increase the lifespan of obese mice and found that the compound reversed nearly all of the changes in gene expression patterns found in mice on high calorie diets—some of which are associated with diabetes, heart disease, and other significant diseases related to obesity.[9]
Numerous in vitro and in vivo studies have demonstrated the positive effects of resveratrol in cardiovascular diseases (CVD). The primary beneficial effects of resveratrol are cardioprotective, anti-hypertensive, vasodilatory, anti-diabetic, and improvement of lipid status.[10]
Stilbene compounds are part of a vast group of natural defense polyphenols occurring in many plant species. Plants biosynthesize their secondary metabolites for specific reasons. Resveratrol is a phytoalexin, a substance produced in response to abiotic stress. This substance exerts antimicrobial and antioxidant activities against phytopathogens and ozone or UV stress. Stilbene compounds can bind several classes of protein structures to elicit a variety of responses.[11]
In healthy mice, resveratrol has been shown to increase lifespan and was protective under conditions of diet-induced obesity. Induction of mitochondrial biogenesis via PGC-1α activation is a major contributor of the beneficial effects of resveratrol.[12],[13],[14]
Mice fed resveratrol had bigger and denser mitochondria amassed between adjacent myofibrils in their nonoxidative fibers. Growth of mitochondria was confirmed by increased mitochondrial size and mtDNA (mitochondrial DNA) content. The max VO2 rate (the maximum capacity to transport and utilize oxygen during incremental exercise) was much higher in the nonoxidative muscle fibers of mice given resveratrol, which means there was an increased oxidative capacity. All these factors imply that resveratrol increases the ratio of oxidative to nonoxidative muscle fibers.
Since resveratrol has the possibility to change muscle fibers to a dominance of oxidative type 1 fibers, it makes sense that this would improve endurance since these types of fibers have a higher resistance to muscle fatigue. Researchers found that mice given resveratrol outran a control group by almost twice the distance. This means that resveratrol greatly increases resistance to muscular fatigue due to increased mitochondrial activation and a conversion of muscle to slow twitch fibers.
In another laboratory study, resveratrol significantly improved exhaustive swimming time and produced dose-dependent decreases in serum lactate and ammonia levels and CK activity after a 15-minute swimming test. The improvement was related to increased energy utilization (as blood glucose) and decreased serum levels of lactate, ammonia, and CK. Therefore, resveratrol could be a potential agent that improves energy transfer with an anti-fatigue pharmacological effect.[15]
The intake of resveratrol, together with habitual exercise, is beneficial for suppressing age-related decline in physical performance. Research indicates that these effects are attributable, at least in part, to improved mitochondrial function in skeletal muscle.[16]
Resveratrol inhibits cardiovascular disease through multiple mechanisms:
- Anti-platelet activity/anti-thrombin: lowers fibrinogen[17]
- Improves endothelial function and inhibits LDL oxidation[18]
- Inhibits platelet aggregation and enhances nitric oxide release[19]
- Inhibits atherosclerosis and vascular inflammation[20],[21]
- Raises GSH, inhibits oxidative endothelial damage[22]
- Improves blood flow in the brain by 30 per cent, reducing the risk of stroke[23]
- Inhibits stroke – reduces MMP-9, a contributor to neuronal injury[24]
- Improves chronic venous insufficiency – edema-protective (ankles and calf)[25]
- Lowers homocysteine[26]
- Potent cardioprotective agent. Cardioprotective effects may be due to a reduction of atrial natriuretic peptide and transforming growth factor-beta1, which are known to protect the heart from detrimental remodeling.[27]
- Suppresses lipid peroxidation and significantly improves oxidative status and release of nitric oxide. Prevents hypertrophic and apoptotic consequences induced by high blood pressure.[28]
Pterostilbene
Pterostilbene is a naturally occurring phytoalexin identified in several plant species, including the well-known Aruvedic herb Pterocarpus marsupium. Although resveratrol is well-known for its health promoting benefits, pterostilbene may be the superior stilbene compound. With respect to mitochondrial genes, studies show that more than 100 genes involved in diverse mitochondrial functions are up-regulated by pterostilbene treatment.
Pterostilbene affects the expression of a diverse group of genes involved in methionine metabolism, response to drug, transcription factor activity, and mitochondrion functions. Additional analyses indicate that many genes involved in lipid metabolism are also affected. The observed response of lipid metabolism genes is in agreement with the known hypolipidemic properties of pterostilbene mediated through the activation of PPARα.[29]
In one study, pterostilbene supplementation was tested on skeletal muscle adaptations to exercise training in rats. After 4 weeks, the pterostilbene supplemented group showed that exercise training resulted in significant increases in time-to-exhaustion, the proportion of slow-twitch fibers, muscular angiogenesis, and mitochondrial biogenesis in rats, and these effects induced by exercise training could be augmented by pterostilbene supplementation. The researchers concluded that pterostilbene promotes skeletal muscle adaptations to exercise training, thereby enhancing endurance capacity.[30]
Creatine
Creatine is a natural substance that improves strength, increases lean muscle mass, and helps the muscles recover more quickly during exercise. The body produces some of the creatine it uses. It also comes from protein-rich foods such as meat or fish, and from supplements. Creatine turns into creatine phosphate in the body, where it helps make adenosine triphosphate (ATP). ATP provides the energy for muscle contractions throughout the body.
Magnesium-creatine chelate supplementation has been shown to improve exercise performance[31] and allow runners to reach their physiological threshold later in exercise.[32]
“Creatine MagnaPower” (MP), a patented magnesium creatine chelate, provides the body with a readily available source of magnesium while also making creatine more active by protecting it from cyclization. This patented mineral amino acid chelate contributes to an overall positive effect on many functions, including a non-steroidal anabolic enhancing effect. Creatine MP also provides rehydration and endurance in times of physiological stress.[33],[34]
As noted above, creatine is essential for normal muscle function. Statins inhibit guanidinoacetate methyl transferase (GAMT), the last enzyme in the synthesis of creatine; thus, they decrease its intracellular content. Such decreased content could cause mitochondrial impairment since creatine is the final acceptor of ATP at the end of mitochondrial oxidative phosphorylation. Accordingly, ATP synthesis is decreased in statin-treated cells.
The myopathy induced by statins is characterized by an increased urinary creatine–creatinine ratio, although muscle-toxicity symptoms and biopsy evidence of mitochondrial dysfunction without creatine kinase elevation have been documented during statin therapy.[35]
There is an extensive and still growing body of literature supporting the efficacy of creatine supplementation.[36] Creatine supplementation prevents the opening of the mitochondrial permeability transition pore that is caused by statins. Clinically, creatine administration prevents statin myopathy in statin-intolerant patients.[37]
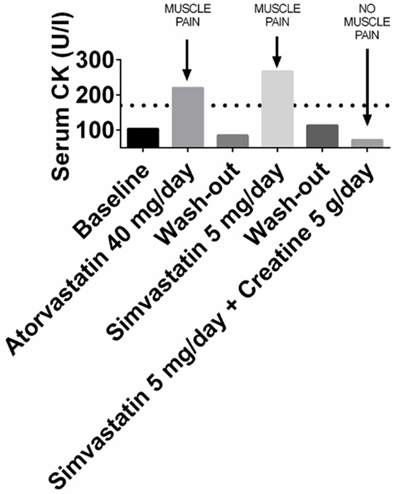
In one study, serum levels of creatine kinase (CK) and muscle pain were treated with creatine supplementation. Muscle pain occurred and CK levels rose to abnormal levels when statins were prescribed, but not when the statin was prescribed together with creatine.[38]
Coenzyme‐Q10 (CoQ10)
Research shows that reduced synthesis of coenzyme Q10 may be the cause of statin related myopathy.[39] There are also indications that statins lead to a reduction of serum CoQ10 levels.[40] Fortunately, CoQ10 supplementation can partially mitigate statin related mitochondrial dysfunction.[41] In fact, the rationale for using CoQ10 to treat statin myalgias is so overwhelming that Merck & Co, Inc. pursued a patent for a CoQ10-statin combination product. They were issued two patents for this product, which was meant to counteract statin-associated myopathy and to reduce the elevated transaminase levels produced by the statin.[42]
Previous studies have demonstrated a possible association between the induction of CoQ10 after statin treatment and statin-induced myopathy. In a meta-analysis of randomized controlled trials investigating the effect of CoQ10 on statin-induced myopathy, researchers evaluated twelve randomized controlled trials with a total of 575 patients; 294 patients were in the CoQ10 supplementation group and 281 were in the placebo group.
Compared with placebo, CoQ10 supplementation ameliorated statin-associated muscle symptoms, such as muscle pain, muscle weakness, muscle cramp, and muscle tiredness, whereas no reduction in the plasma creatine kinase level was observed after CoQ10 supplementation.
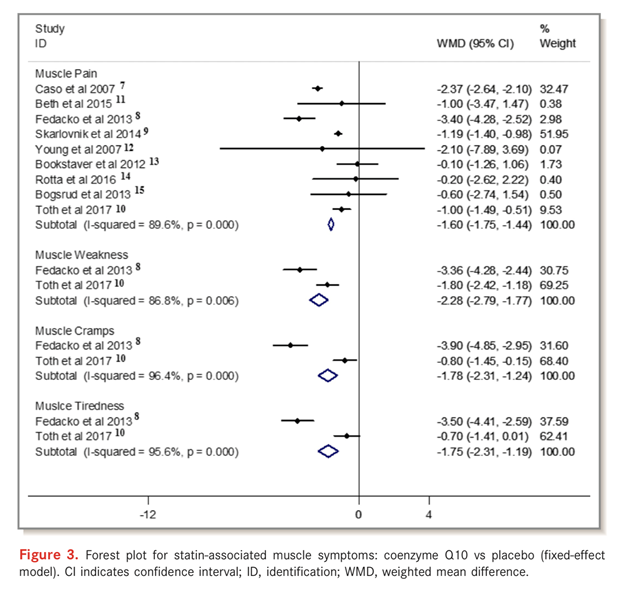
The study concluded that CoQ10 supplementation ameliorated statin-associated muscle symptoms, implying that CoQ10 supplementation may be a complementary approach to manage statin-induced myopathy.[43]
Stabilized R-lipoic acid
Stabilized R-lipoic acid has been shown to reduce age-related mitochondria oxidative damage,[44] lower glucose and lactate,[45] improve metabolic activity, and lower oxidative stress and oxidative damage.[46]
BioEnhanced™ Na-RALA is a stabilized form of RLA that does not degrade at high temperatures, is more bioavailable than regular RLA, and has no solvent residues. RLA can be converted, or reduced, to DHLA, or dihydrolipoic acid. These two forms of LA make up a “redox couple,” which means that each form can chemically change, or donate back and forth.
Clinical studies have shown that the use of stabilized R-lipoic acid works together with CoQ10, improving quality of life by preventing free radical damage to proteins.[47]
Disturbances in acid-base balance, such as acidosis and alkalosis, have the potential to alter the pharmacologic and toxicologic outcomes of statin therapy.[48]
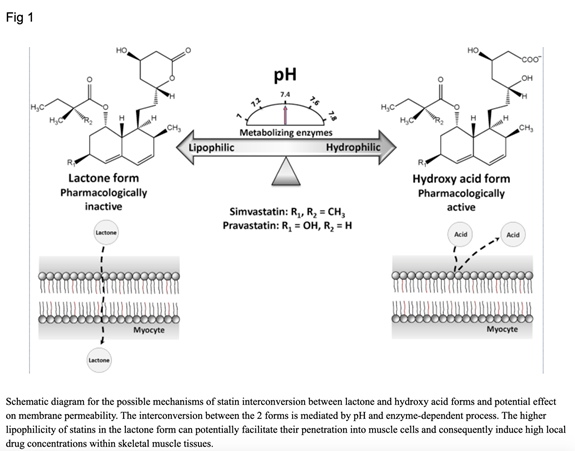
Other studies have shown that statins may activate muscle atrophy genes, may increase muscle specific NO‐synthetase and NO production, and may decrease fatty acid oxidation.[49],[50]
Patients experiencing muscle pains on statins were 11-times more likely to be a heterozygous carrier for the carnitine palmitoyltransferase-2 deficiency, and 31% of muscle biopsies evaluated had carnitine abnormalities. Moreover, patients were 20-times more likely to be carriers for McArdle’s disease (a glycogen storage disease), and a third had lipid storage problems. This trial suggests that people who experience muscle pains on a statin are more likely to have an underlying metabolic muscle disease with the symptoms of statin muscle pain being brought out in these carriers. Furthermore, almost 50% of the analyzed samples had CoQ10 levels that were 2–4 standard deviations below normal. Therefore, supplementing with both CoQ10 and l-carnitine may be a rational approach to treating certain statin myalgias.[51]
Some patients are susceptible to statin-induced myopathy due to variations in genes encoding proteins involved in statin uptake and biotransformation such as the solute carrier organic anion transporter family member 1B1 (SLCO1B1) or cytochrome P450 (CYP2D6, CYP3A4, CYP3A5). Carriers for carnitine palmitoyltransferase II deficiency and McArdle disease also present with higher prevalence of statin-induced myopathy.[52]
Vitamin D
A recent study evaluated whether vitamin D status modifies the association between statin use and musculoskeletal pain in a sample representative of the general population. In the study, musculoskeletal symptoms and statin use were self-reported. Vitamin D status was assessed using serum 25 hydroxyvitamin D (25[OH]D), categorized as <15 ng/mL or ≥15 ng/mL. To evaluate if vitamin D status modifies the association between statin use and prevalent musculoskeletal pain, the researchers performed multivariable-adjusted logistic regression models stratified by 25(OH)D status.
Among 5907 participants ≥40 years old, mean serum 25(OH)D was 23.6 ng/mL. In stratified multivariable-adjusted logistic regression models, individuals with 25(OH)D <15 ng/mL using a statin had significantly higher odds of musculoskeletal pain compared to those not using a statin. Among those with 25(OH)D ≥15 ng/mL, there was no significant association between statin use and musculoskeletal pain.
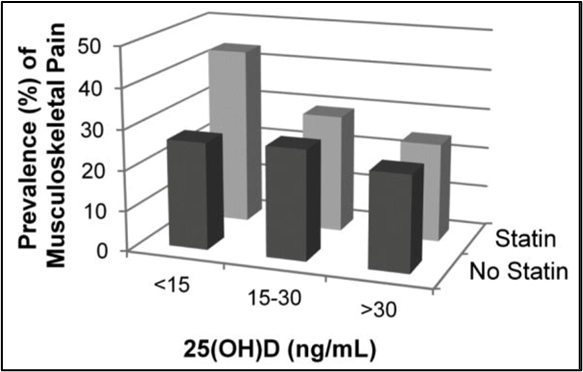
The study concluded that among adults ≥ 40 years old with 25(OH)D <15 ng/mL, statin users had nearly 2 times greater odds of reporting musculoskeletal pain compared to non-statin users. This supports the hypothesis that vitamin D deficiency modifies the risk of musculoskeletal symptoms experienced with statin use.[53]
Supplementing with Vitamin D
- It’s best to supplement with a food grown form of vitamin D that also includes a small amount of vitamin K (K-1 or K-2, or both), and vitamin A. Vitamin D grown in saccharomyces cerevisiae, creates a vitamin D food-like matrix and contains various immune-enhancing compounds such as β-glucan, nucleic acids, mannan oligosaccharides and chitin. This has been proven to enhance immune response and growth.
- Simply taking extra vitamin D is not the correct approach. Instead, the appropriate amount must be provided to maintain serum levels in a healthy range of 25 OH (>45 <80).
- Always check both forms of vitamin D; that means 25-OH and 1,25 dihydroxyvitamin (di OH). While 1,25 vitamin D is the most potent vitamin D metabolite, 25-OH vitamin D levels more accurately reflect vitamin D status because parathyroid hormone and other hormones do not influence it. If you only check 25-OH after supplementing with vitamin D, and the levels don’t increase, proceed by checking the 1,25 di OH level and the parathyroid hormone level. If these are either high normal, or elevated out-of-range, then you need to seek a professional skilled at normalizing vitamin D levels.
Curcumin
Curcumin is the polyphenolic ingredient of Curcuma longa, which has numerous pharmacological properties against a vast range of diseases. Curcumin has several mechanisms of actions relevant to the treatment of SIM. These include the capacity to prevent and reduce delayed onset muscle soreness by blocking the nuclear factor inflammatory pathway, attenuation of muscular atrophy, enhancement of muscle fiber regeneration following injury, and analgesic and antioxidant effects.
Curcumin can also increase the levels of cyclic adenosine monophosphate, which leads to an increase in the number of mitochondrial DNA duplicates in skeletal muscle cells. Finally, because of its essential lipid-modifying properties, curcumin is a possible adjunct to statin therapy in patients with SIM, assisting in lowering of low-density lipoprotein cholesterol and potentially reducing the dose of statins.[54]
Cistanche (Herba Cistanches)
Cistanche is a parasitic plant that is commonly is used in Traditional Chinese Medicine (TCM) for treating kidney deficiency, impotence, female infertility and Age-related constipation. Recent research suggests that cistanche may be protective against simvastatin-induced muscle toxicity.[55],[56]
Yang xin shi
The Yang xin shi tablet (YXST) is prescribed in TCM for improving the clinical symptoms of patients with cardiovascular diseases in combination with conventional drugs.
YXST Ingredients

YXST can effectively inhibit the myolysis induced by the statin drug Simvastatin. Mitochondrial activities are closely related to energy production. In studies, YXST was shown to significantly increase the activity of mitochondrial complex III and glycogen utilization to counteract the side effects of statin in the skeletal muscle.[57]
Potassium Bicarbonate
Research indicates that potassium bicarbonate improves endothelial function, cardiovascular risk factors, and bone turnover in mild hypertensives.[58]
Supplementing with potassium bicarbonate has been shown to prevent statin-induced myopathy and further demonstrates a preventive effect on statin (cerivastatin)-induced increase in CPK concentrations.[59]
Choosing a Natural Approach
Achieving healthy cholesterol levels can generally be accomplished through attention to a healthy Mediterranean diet, regular exercise, and appropriate supportive supplements, such as those discussed here.
Because of unique individual differences, optimizing cholesterol levels may require consultation with a health care professional skilled in the use of nutritional and botanical supplements.
God embraces you as you are—shadow and light, everything. God embraces it, by grace. —Brother David Steindl-Rast
References
[1] Chermnykh NS, Shimanovskiĭ NL, Shutko GV, Syrov VN. Deĭstvie metandrostenolona i ékdisterona na fizicheskuiu vynoslivost’ zhivotnykh i obmen belkov v skeletnykh myshtsakh [The action of methandrostenolone and ecdysterone on the physical endurance of animals and on protein metabolism in the skeletal muscles]. Farmakol Toksikol. 1988 Nov-Dec;51(6):57-60. Russian. PMID: 3234543.
[2] Dinan L, Dioh W, Veillet S, Lafont R. 20-Hydroxyecdysone, from Plant Extracts to Clinical Use: Therapeutic Potential for the Treatment of Neuromuscular, Cardio-Metabolic and Respiratory Diseases. Biomedicines. 2021 Apr 29;9(5):492. doi: 10.3390/biomedicines9050492. PMID: 33947076; PMCID: PMC8146789.
[3] Parr MK, Botrè F, Naß A, Hengevoss J, Diel P, Wolber G. Ecdysteroids: A novel class of anabolic agents? Biol Sport. 2015 Jun;32(2):169-73. doi: 10.5604/20831862.1144420. Epub 2015 Mar 15. PMID: 26060342; PMCID: PMC4447764.
[4] Cahlíková L, Macáková K, Chlebek J, Host’álková A, Kulhánková A, Opletal L. Ecdysterone and its activity on some degenerative diseases. Nat Prod Commun. 2011 May;6(5):707-18. PMID: 21615037.
[5] Sharipov RR, Kotsiuruba AV, Kop”iak BS, Sahach VF. [Induction of oxidative stress in heart mitochondria in brain focal ischemia-reperfusion and protective effect of ecdysterone]. Fiziol Zh. 2014;60(3):11-7. Ukrainian. PMID: 25095666.
[6] Niranjan Das, Siddhartha Kumar, Mishra, Anusha Bishayee et. al., The phytochemical, biological, and medicinal attributes of phytoecdysteroids: an updated review, Acta Pharmaceutica Sinica B Available online 16 October 2020, https://doi.org/10.1016/j.apsb.2020.10.012
[7] Peschel W, Kump A, Prieto JM. Effects of 20-hydroxyecdysone, Leuzea carthamoides extracts, dexamethasone and their combinations on the NF-κB activation in HeLa cells. J Pharm Pharmacol. 2011 Nov;63(11):1483-95. doi: 10.1111/j.2042-7158.2011.01349.x. PMID: 21988429.
[8] Kokoska L, Janovska D. Chemistry and pharmacology of Rhaponticum carthamoides: a review. Phytochemistry. 2009 May;70(7):842-55. doi: 10.1016/j.phytochem.2009.04.008. Epub 2009 May 18. PMID: 19457517.
[9] Harvard Medical School. “Red Wine Molecule Extends Lifespan Of Fat Mice Lives By Reversing Obesity-Related Gene Pathways.” ScienceDaily. ScienceDaily, 2 November 2006. <www.sciencedaily.com/releases/2006/11/061101151156.htm
[10] Gligorijević N, Stanić-Vučinić D, Radomirović M, Stojadinović M, Khulal U, Nedić O, Ćirković Veličković T. Role of Resveratrol in Prevention and Control of Cardiovascular Disorders and Cardiovascular Complications Related to COVID-19 Disease: Mode of Action and Approaches Explored to Increase Its Bioavailability. Molecules. 2021 May 11;26(10):2834. doi: 10.3390/molecules26102834. PMID: 34064568.
[11] Mika Reinisalo,1 Anna Kårlund,2 Ali Koskela,, Kai Kaarniranta,1,3 and Reijo O. Karjalainen, Polyphenol Stilbenes: Molecular Mechanisms of Defense against Oxidative Stress and Aging-Related Diseases, Oxidative Medicine and Cellular Longevity Volume 2015, Article ID 340520, 24 pages http://dx.doi.org/10.1155/2015/340520
[12] Baur JA, Pearson KJ, Price NL, Jamieson HA, Lerin C, Kalra A et al. (2006). Resveratrol improves health and survival of mice on ahigh-calorie diet. Nature 444: 337–342.
[13] Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F et al. (2006). Resveratrol improves mitochondrialfunction and protects against metabolic disease by activating SIRT1and PGC-1alpha. Cell 127: 1109–1122.
[14] Pearson KJ, Baur JA, Lewis KN, Peshkin L, Price NL, Labinskyy N et al. (2008). Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending lifespan. Cell Metab 8: 157–168.
[15] Wu RE, Huang WC, Liao CC, Chang YK, Kan NW, Huang CC. Resveratrol protects against physical fatigue and improves exercise performance in mice. Molecules. 2013 Apr 19;18(4):4689-702. doi: 10.3390/molecules18044689. PMID: 23603951; PMCID: PMC6270062.
[16] Murase T, Haramizu S, Ota N, Hase T. Suppression of the aging-associated decline in physical performance by a combination of resveratrol intake and habitual exercise in senescence-accelerated mice. Biogerontology. 2009 Aug;10(4):423-34. doi: 10.1007/s10522-008-9177-z. Epub 2008 Oct 1. PMID: 18830683.
[17] Zbikowska HM. Olas B Prof. H.M. Zbikowska, Antioxidants with carcinostatic activity (resveratrol, vitamin E and selenium) in modulation of blood platelet adhesion. Vol 51(3) (pp 513-520), 2000.
[18] Kiesewetter, Dr. H. Good clinical practice in accordance with clinical studies on the efficacy and tolerability of red wine leaf extract AS 195 in chronic venous insufficiency. Vasomed. Vol 14(1) (pp 18-20), 2002.
[19] Teixeira S. J Orthop Sports Phys Ther 2002;32:357-363.
[20] Wang Z, Zou J, Cao K, Hsieh TC, Huang Y, Wu JM. Dealcoholized red wine containing known amounts of resveratrol suppresses atherosclerosis in hypercholesterolemic rabbits without affecting plasma lipid levels. Int J Mol Med. 2005 Oct;16(4):533-40.
[21] Rimando AM, Kalt W, Magee JB, Dewey J, Ballington JR. Resveratrol, pterostilbene, and piceatannol in vaccinium berries. J Agric Food Chem. 2004 Jul 28;52(15):4713-9
[22] Ruf JC. Alcohol, wine and platelet function. Biol Res. 2004;37(2):209-15.
Freedman, Jane, Drinking Concord grape juice significantly increased good cholesterol and significantly lowered two markers of inflammation in people with stable coronary artery disease, November, 2004 Journal Arteriosclerosis, Thrombosis and Vascular Biology, Boston University School of Medicine.
[23] Lu KT, Chiou RY, Chen LG, Chen MH, Tseng WT, Hsieh HT, Yang YL. Neuroprotective effects of resveratrol on cerebral ischemia-induced neuron loss mediated by free radical scavenging and cerebral blood flow elevation.
[24] J Agric Food Chem. 2006 Apr 19;54(8):3126-31. Gao D, Zhang X, Jiang X, Peng Y, Huang W, Cheng G, Song L. Resveratrol reduces the elevated level of MMP-9 induced by cerebral ischemia-reperfusion in mice. Life Sci. 2006 Apr 25;78(22):2564-70.
[25] Kiesewetter, Dr. H. Good clinical practice in accordance with clinical studies on the efficacy and tolerability of red wine leaf extract AS 195 in chronic venous insufficiency. Vasomed. Vol 14(1) (pp 18-20), 2002. Universitatsklinikum Charite[spacing acute], Institut fur Transfusionsmedizin, Schumannstra[latin sharp s]e 20-21, 10117
[26] Schroecksnadel K, Winkler C, Wirleitner B, Schennach H, Weiss G, Fuchs D. Anti-inflammatory compound resveratrol suppresses homocysteine formation in stimulated human peripheral blood mononuclear cells in vitro. Clin Chem Lab Med. 2005;43(10):1084-8.
[27] Lin SM, Chih CL, Nien MW, Su HH, Hu BR, Huang SS, Tsai SK. Resveratrol reduces infarct size and improves ventricular function after myocardial ischemia in rats. Lin JF, Life Sci. 2008 Aug 29;83(9-10):313-7. Epub 2008 Jun 27.
[28] Grujić-Milanović J, Jaćević V, Miloradović Z, Jovović D, Milosavljević I, Milanović SD, Mihailović-Stanojević N. Resveratrol Protects Cardiac Tissue in Experimental Malignant Hypertension Due to Antioxidant, Anti-Inflammatory, and Anti-Apoptotic Properties. Int J Mol Sci. 2021 May 8;22(9):5006. doi: 10.3390/ijms22095006. PMID: 34066865.
[29] Zhiqiang Pan, Ameeta K Agarwal, Tao Xu, Qin Feng, Scott R Baerson, Stephen O Duke, and Agnes M Rimando, Identification of molecular pathways affected by pterostilbene, a natural dimethylether analog of resveratrol, BMC Med Genomics. 2008; 1: 7. Published online 2008 March 20. doi: 10.1186/1755-8794-1-7.
[30] Zheng J, Liu W, Zhu X, Ran L, Lang H, Yi L, Mi M, Zhu J. Pterostilbene Enhances Endurance Capacity via Promoting Skeletal Muscle Adaptations to Exercise Training in Rats. Molecules. 2020 Jan 2;25(1):186. doi: 10.3390/molecules25010186. PMID: 31906449; PMCID: PMC6982856.
[31] Brilla, L.R. Giroux, M. Taylor, A. Knutzen, K., Magnesium-Creatine chelate supplementation may allow runners to reach their physiological threshold later in exercise, Nutrition Week 2002 supplement to the February issue of the American Journal of Clinical Nutrition (75, 2(S), 2002) (www.ajcn.org).
[32] Selsby JT1, DiSilvestro RA, Devor ST. Mg2+-creatine chelate and a low-dose creatine supplementation regimen improve exercise performance, J Strength Cond Res. 2004 May;18(2):311-5.
[33] Brilla, L.R., FACSM, Giroux, M.S. Taylor, A. Kennedy, J. Ramierex, R.E. Puz, D. and Knutzen, K.M. FACSM. Magnesium-Creatine Supplementation on Total Body Water, ICF and ECF Compartments, Western Washington University, WA. American College Sports Medicine 2002.
[34] Brilla, L.R. Kennedy, J.S. Knutzen K.M. Magnesium-Creatine Supplementation Effects of Exhaustive Exercise, Western Washington University, Bellingham, WA. Am J Clin Nutr, V75, 2 (S) Feb 2002 ISSN 0002-9165.
[35] Shewmon D.A., Craig J.M. Creatine supplementation prevents statin-induced muscle toxicity. Ann. Internal Med. 2010;153:690–692. doi: 10.7326/0003-4819-153-10-201011160-00024.
[36] Gualano B, Roschel H, Lancha-Jr AH, Brightbill CE, Rawson ES. In sickness and in health: the widespread application of creatine supplementation. Amino Acids. 2011 Nov 19.
[37] Balestrino M, Adriano E. Creatine as a Candidate to Prevent Statin Myopathy. Biomolecules. 2019 Sep 17;9(9):496. doi: 10.3390/biom9090496. PMID: 31533334; PMCID: PMC6770148.
[38] Balestrino M., Adriano E. Statin-induced myopathy prevented by creatine administration. BMJ Case Rep. 2018;2018 doi: 10.1136/bcr-2018-225395.
[39] Nielsen ML, Pareek M, Henriksen JE. Nedsat koenzym Q10 kan være årsag til statinassocieret myopati [Reduced synthesis of coenzyme Q10 may cause statin related myopathy]. Ugeskr Laeger. 2011 Nov 14;173(46):2943-8. Danish. PMID: 22094213.
[40] Banach M, Serban C, Ursoniu S, Rysz J, Muntner P, Toth PP, Jones SR, Rizzo M, Glasser SP, Watts GF, Blumenthal RS, Lip GY, Mikhailidis DP, Sahebkar A, Lipid and Blood Pressure Meta‐analysis Collaboration (LBPMC) Group, Statin therapy and plasma coenzyme Q10 concentrations – a systematic review and meta‐analysis of placebo‐controlled trials. Pharmacol Res 2015;99:329–336.
[41] Langsjoen PH, Langsjoen JO, Langsjoen AM, Lucas LA. Treatment of statin adverse effects with supplemental Coenzyme Q10 and statin drug discontinuation. Biofactors. 2005;25(1-4):147-52
[42] CoQ10 and L-carnitine for Statin Myalgia? – Medscape – Oct 01, 2012. Expert Rev Cardiovasc Ther. 2012;10(10):1329-1333. © 2012 Expert Reviews Ltd.
[43] Qu H, Guo M, Chai H, Wang WT, Gao ZY, Shi DZ, Effects of Coenzyme Q10 on Statin-Induced Myopathy: An Updated Meta-Analysis of Randomized Controlled Trials, J Am Heart Assoc. 2018 Oct 2;7(19):e009835. doi: 10.1161/JAHA.118.009835.
[44] Bharat S, Cochran BC, Hsu M, Liu J, Ames BN, Andersen JK.Pre-treatment with R-lipoic acid alleviates the effects of GSH depletion in PC12 cells: implications for Parkinson’s disease therapy. Neurotoxicology. 2002 Oct;23(4-5):479-86.
[45] Korotchkina LG, Sidhu S, Patel MS. R-lipoic acid inhibits mammalian pyruvate dehydrogenase kinase. Free Radic Res. 2004 Oct;38(10):1083-92.
[46] Hagen TM, Ingersoll RT, Lykkesfeldt J, Liu J, Wehr CM, Vinarsky V, Bartholomew JC, Ames AB. (R)-alpha-lipoic acid-supplemented old rats have improved mitochondrial function, decreased oxidative damage, and increased metabolic rate. FASEB J. 1999 Feb;13(2):411-8.
[47] Bharat S, Cochran BC, Hsu M, Liu J, Ames BN, Andersen JK.Pre-treatment with R-lipoic acid alleviates the effects of GSH depletion in PC12 cells: implications for Parkinson’s disease therapy. Neurotoxicology. 2002 Oct;23(4-5):479-86. Sethumadhavan S, Chinnakannu P. Carnitine and lipoic acid alleviates protein oxidation in heart mitochondria during aging process. Biogerontology. 2006; Apr;7(2):101-9.).
[48] Taha DA, De Moor CH, Barrett DA, et al. The role of acid-base imbalance in statin-induced myotoxicity. Transl Res. 2016;174:140-160.e14. doi:10.1016/j.trsl.2016.03.015
[49] Goodman CA, Pol D, Zacharewicz E, Lee‐Young RS, Snow RJ, Russell AP, McConell GK. Statin‐induced increases in atrophy gene expression occur independently of changes in PGC1α protein and mitochondrial content. PLoS One 2015;10:e0128398
[50] Finsterer J, Frank M. Management of statin myopathy. J Cachexia Sarcopenia Muscle. 2017 Jun;8(3):512-513. doi: 10.1002/jcsm.12207. Epub 2017 May 2. PMID: 28466578; PMCID: PMC5476849.
[51] Vladutiu G, Isackson P, Wortmann R et al. Metabolic muscle disorders and cholesterol-lowering drugs. Abstracts of: The American College of Rheumatology 2004 meeting. San Antonio, TX, USA, 16–21 October 2004 (Abstract 1784).
[52] Apostolopoulou M, Corsini A, Roden M. The role of mitochondria in statin-induced myopathy. Eur J Clin Invest. 2015 Jul;45(7):745-54. doi: 10.1111/eci.12461. Epub 2015 Jun 15. PMID: 25991405.
[53] Morioka TY, Lee AJ, Bertisch S, Buettner C. Vitamin D status modifies the association between statin use and musculoskeletal pain: a population based study. Atherosclerosis. 2015 Jan;238(1):77-82. doi: 10.1016/j.atherosclerosis.2014.11.012. Epub 2014 Nov 20. PMID: 25437894; PMCID: PMC4277894.
[54] Sahebkar A, Saboni N, Pirro M, Banach M. Curcumin: An effective adjunct in patients with statin-associated muscle symptoms? J Cachexia Sarcopenia Muscle. 2017 Feb;8(1):19-24. doi: 10.1002/jcsm.12140. Epub 2016 Sep 22. PMID: 27897416; PMCID: PMC5326825.
[55] Wat E, Ng CF, Koon CM, Wong EC, Tomlinson B, Lau CB. The protective effect of Herba Cistanches on statin-induced myotoxicity in vitro. J Ethnopharmacol. 2016 Aug 22;190:68-73. doi: 10.1016/j.jep.2016.06.020. Epub 2016 Jun 7. PMID: 27286913.
[56] Wat E, Ng CF, Koon CM, Zhang C, Gao S, Tomlinson B, Lau CBS. The adjuvant value of Herba Cistanches when used in combination with statin in murine models. Sci Rep. 2017 Aug 24;7(1):9391. doi: 10.1038/s41598-017-10008-7. PMID: 28839280; PMCID: PMC5570940.
[57] Wu RM, Jiang B, Li H, et al. Yang xin shi tablet enhances adaptability to exercise training by relieving statin-induced skeletal muscle injury. Chin Med J (Engl). 2020;133(18):2266-2268. doi:10.1097/CM9.0000000000001028
[58] He FJ, Marciniak M, Carney C, Markandu ND, Anand V, Fraser WD, Dalton RN, Kaski JC, MacGregor GA. Effects of potassium chloride and potassium bicarbonate on endothelial function, cardiovascular risk factors, and bone turnover in mild hypertensives. Hypertension. 2010 Mar;55(3):681-8. doi: 10.1161/HYPERTENSIONAHA.109.147488. Epub 2010 Jan 18.
[59] Kobayashi M, Kagawa T, Narumi K, Itagaki S, Hirano T, Iseki K. Bicarbonate supplementation as a preventive way in statins-induced muscle damage. J Pharm Pharm Sci. 2008;11(1):1-8. doi: 10.18433/j33018. PMID: 18445360.