I have been involved in the health industry for four decades and in clinical practice for three decades, and have seen every possible variation of supposedly health-promoting diet come and go. Macrobiotic, raw food, fat free, vegan, and high protein diets have been touted as diets for preventing or healing from cancer, most of them offering up a confusing array of contradictory advice. The most recent diet to appear on the scene is the ketogenic (keto) diet, a high fat and low protein regime virtually devoid of carbohydrates. I would like to share my opinion on why I am not in favor of the ketogenic diet in general and the very rare and specific circumstances in which it could possibly have benefit with short-term use in people with brain cancer.

A keto diet is high fats, moderate in protein, and extremely low in all carbohydrates (both good and bad). By restricting all (including healthy) carbohydrate consumption to a mere 20-50g per day the keto diet aims to starve the cell of body (including the brain) of glucose. When glucose stores are depleted due to starvation or extremely low carbohydrate consumption, the body goes into ketosis (this is where the name keto diet comes from). In ketosis, the liver breaks fat (and secondary protein) down into ketone bodies as a secondary fuel source for the brain. While this diet, by inducing fat breakdown via ketosis can produce impressive weight loss, this “quick fix” can also come at a cost.
Why Have Keto Diets Become Popular?
The highly restrictive ketogenic diet has gained popularity primarily because it promises weight loss. With so many people struggling to lose weight, it’s easy to understand why people are attracted to a diet that allows unlimited amounts of fatty foods like bacon, eggs, and burgers (but hold the bun).
Low-carb diets like the keto diet do appear to lead to short-term weight loss, but they’re not significantly more effective than any other commercial or self-help diet. In a comparison of various popular diets published in the Journal of the American Medical Association in 2014,1 researchers analyzed 48 separate diets in which participants were randomly assigned to one of several diets.
The diets included the Atkins (a low-carb diet), South Beach and Zone diets as well as very low-fat diets like the Ornish diet and portion-control diets like Jenny Craig and Weight Watchers. The results showed that any of these diets resulted in more weight loss than no diet at all after six months. Low-fat and low-carb diets were pretty much indistinguishable, with low-carb dieters losing 19 pounds on average, and low-fat dieters losing an average of 17.6 pounds, both compared to non-dieters. At 12 months, the benefits showed signs of leveling of for both types of diets, with both low-fat and low-carb dieters reporting being 16 pounds lighter, on average, than non-dieters.
The biggest problem is that the ketogenic diet severely restricts carbohydrates, including all whole grains, legumes, most fruits, and starchy vegetables. Restricting or eliminating these foods makes it extremely difficult to obtain the micronutrients we need for optimal health.
Why I am Opposed to the Ketogenic Diet
The keto diet is also promoted as a potential cancer treatment, without significant evidence for its effectiveness and no long-term data to support its use. I have seen many patients steered to this diet with devastating results. A patient of mine who had been living well with metastatic breast cancer decided to go off the health-supportive program that had kept her well for fifteen years, and switched to a regime that included the keto diet. I understand her desire for a cure, but the result was disastrous. Her cancer began to grow uncontrollably, leading to her death within less than three months. I witnessed the same occur with an ovarian cancer patient.
In my opinion, based on my years of careful research and clinical practice, keto diets are not appropriate or optimal for promoting long-term health, nor are keto diets a “cure” for cancer. This diet simply has too many significant drawbacks, not the least of which is that it is difficult to adhere to and not enjoyable for most people. For true nourishment, you must feed your spirit as well as your body, and that includes eating healthful foods that you find appealing.
There is no single dietary villain or savior – a healthy diet is a balancing act of countless different foods and nutrients, including whole food carbohydrates. Eliminating all carbohydrates and going on an extremely high fat diet does not and cannot cure cancer. While I completely agree with eliminating refined, processed carbohydrates, I do not agree with eliminating unrefined, whole food, complex carbohydrates. In some cases, a low-carbohydrate, high-fat diet may lead to an improvement in health, but that’s often because the person was eating a terribly unhealthy diet, and any change toward whole foods (even a ketogenic diet) can be beneficial, at least in the short term.
A diet based on macro-food alterations—a diet extremely high in fat and virtually devoid of carbohydrates—is contrary to everything I know to be true about a healthful diet. It also goes against the wisdom of traditional dietary patterns and the large body of scientific data that supports a balanced whole-foods diet.
In Some Circumstances, a Short-term Ketogenic Diet May Be Useful
Although I am clearly not a proponent of the ketogenic diet, I agree with Ben Franklin, who said, ”a place for everything, everything in its place.” For me, this means that for a brief period in a very specific situation, the ketogenic diet might actually be appropriate for a cancer patient. An instance of this is when a tumor is highly glycolytic and producing a large amount of lactic acid.
It’s important to understand that while all cancers are alike, they are alike in unique ways, and are affected by both the microenvironment and the host. Cancer cells are expert at adapting, and a ketogenic diet has the potential to stimulate cancer cells to adapt into a more aggressive cancer.
Cancer cells need food to survive and grow, and their favorite food is glucose. Compared to healthy cells, cancer cells demonstrate a 3 to 5-fold increase in glucose uptake. It seems simple—deprive cancer cells of glucose, and they’ll die. But research hasn’t proven this to be true.
There’s no question that improving blood sugar levels and lowering A1C, insulin, and leptin are essential for inhibiting cancer, reducing cancer growth, preventing cancer reoccurrence, improving the effectiveness of other therapies, and enhancing lifespan. But a whole food, diverse diet that is mostly plant-based, along with a healthy lifestyle, will accomplish these goals better than a keto diet. I have used this approach with hundreds of patients, and find it effective not only for supporting physical health, but also find that people can embrace and enjoy this way of eating, unlike the keto diet, which is extremely difficult to sustain.
In a 2013 study, researchers discovered that the protein PKCz is responsible for this paradox. The research suggests that glucose depletion therapies might work, as long as the cancer cells produce PKCz. But without PKCz around to keep them addicted to glucose, these tumors create a new metabolic pathway. This altered metabolism helps PKCz-deficient cancer cells survive.2
A keto diet takes advantage of a major metabolic difference between cancerous cells and healthy cells. Most cancerous cells (but not always, and not exclusively) use glucose to produce energy, and they have less metabolic flexibility to use ketones when glucose is unavailable (but they figure it out). A keto diet significantly lowers blood glucose, insulin, and IGF-1 signaling, which also can drive cancer cell proliferation. In a September 2017 review of all clinical trials to date on the keto diet for advanced cancer patients, some studies have shown the keto diet impedes tumor growth, but other studies have not found the same positive results.3
There is some supportive data on short-term keto diets potentially being useful, specifically in the treatment of brain tumors. But this is in comparison to a general “eat what you want” unhealthy Western diet, not a whole food diet that I would recommend based on the principles of the Eclectic Triphasic Medical System (ETMS). There are also real concerns with tumor growth from a keto diet, particularly with certain cancers and specific metabolic mutations.
There is one new proposed biomarker that might provide a clue as to when a short-term keto diet might be useful for a cancer patient. Transketolase-like-1 (TKTL1) is a novel tumor marker associated with aerobic glycolysis of tumor cells. Tumors that correlate with high TKTL1 expression appear to be more aggressive and may require a higher level of aerobic glycolysis. In one study, cancer patients adhering strictly to a keto diet experienced a corresponding drop in TKTL-1. Again, this diet was not compared to a whole foods ETMS diet.4
A Keto Diet Restricts Health-Promoting, Cancer-Suppressing Foods
Complex carbohydrates in the form of fruits, vegetables, potatoes, and whole grains do not cause or promote cancer. In fact, these foods are cancer protective, can contribute to epigenetic reprogramming, and can even suppress growth in existing tumors.
To compare a keto diet to a typical Western diet and think this constitutes the best diet for cancer patients is incorrect. Removing refined sugars and starches is key to a healthy diet, but rather than replacing these cancer-promoting foods with a surplus of fats, replace them with healthful whole grains, fruits, and vegetables. Concentrate on eating a balanced diet that features foods that either target the constitution, the presenting symptoms and nutritional needs, or targets the specific cancer pathways, such as HER2 neu, or gene mutations.
The very low carbohydrate content of a keto diet restricts important fiber-rich, anti-cancer suppressing foods and phytonutrient richness. Phytochemicals in whole foods—particularly vegetables, fruits, and whole grains—have anti-microbial, anti-cancer and cardio-protective benefits and also enhance our immune defenses. The ETMS diet provides powerful longevity-promoting effects, can be tailored to individual tastes, and is a diet that everyone can enjoy.
For the most powerful health protective effects, complex carbohydrates must be eaten as whole foods. There is a synergy of complex phytonutrients in whole grains, fruits and other complex carbohydrates that contribute to their cancer suppressing effects. Refine them, and you turn a healthy food into an unhealthy food.5
For example, hyaluronan oligosaccharides, present in many complex carbohydrates, inhibit anchorage-independent growth of tumors by suppressing the phosphoinositide-3-kinase/Akt cell survival pathway.6-8
In another interesting study, animals with cancerous tumors fed a diet of black rice, a complex carbohydrate, experienced tumor regression by up to 35%. According to the researchers, tumor inhibition was related to a variety of immune enhancing factors, including increases in activity of natural killer cells and increases in released tumor necrosis factor-α, IL-1β, and IL-6 from macrophages; an improvement in the ability of leukocytes to enter the tumor; and a reduction in angiogenesis of the tumor related to reduced COX-2 and 5-LOX expression.9
And in yet another study, beetroot-carrot juice, a sweet juice forbidden in a keto diet, was used as an adjuvant to the antileukemic drug chlorambucil in treating a patient with chronic lymphocytic leukemia. This phytonutrient-rich juice, taken daily, was found to be an effective treatment for CLL alone or in combination with chlorambucil.10
There are now dozens of human studies, including several meta-analyses, that conclude the consumption of whole grains is associated with a significant reduction in both cancer and all-cause mortality. One recent November 2017 published meta-analysis of 19 studies found whole grain consumption was associated with an average 9% reduction in all-cause mortality.11
Digging More Deeply Into the Dangers of a Keto Diet for Cancer
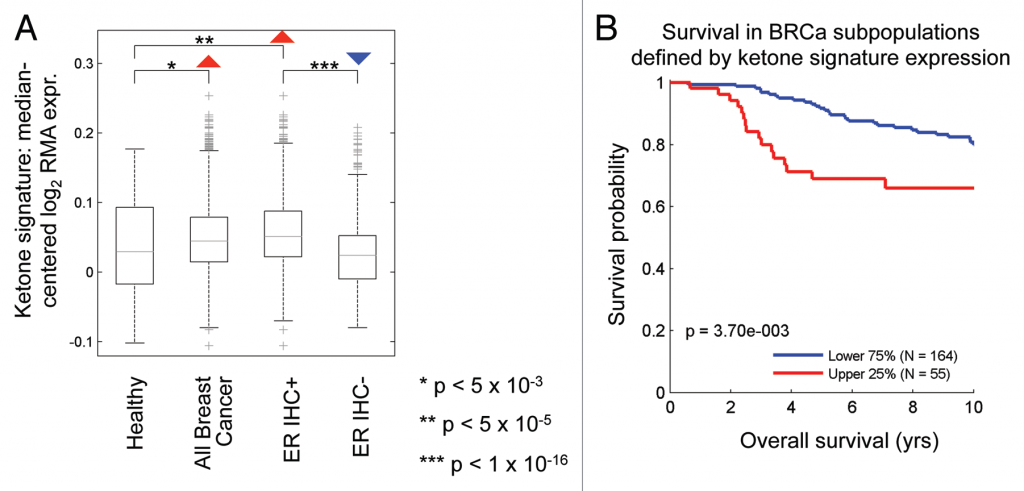
Recent research indicates that ketones and lactate (metabolic byproducts of the keto diet) increase cancer cell “stemness,” which drives recurrence, metastasis, and poor clinical outcomes. In an experimental laboratory study on breast cancer, researchers found that lactate and ketones appear to drive oxidative mitochondrial metabolism in breast cancer cells. This means that clinical outcome in breast cancer could be determined by epigenetics (changes in genes triggered by diet and lifestyle) and energy metabolism, rather than by specific classical gene mutations.
The ketone-induced gene signature is associated with ER(+) breast cancer and predicts poor clinical outcome.12
Emerging Roles of Lipid Metabolism in Cancer Metastasis
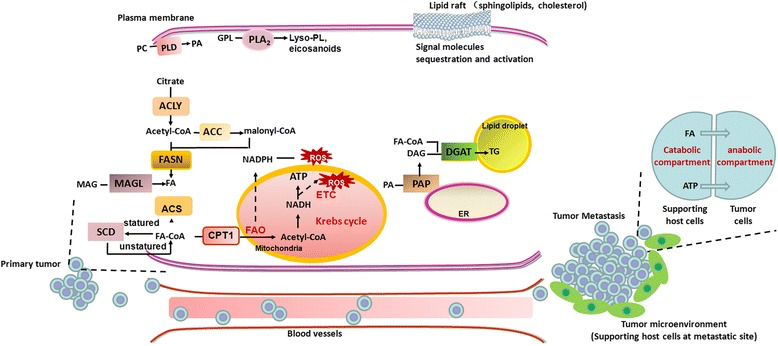
Rewiring of metabolic programs, such as aerobic glycolysis and increased glutamine metabolism, are crucial for cancer cells to shed from a primary tumor, overcome nutrient and energy deficits, and survive and form metastases. In recent studies, researchers have noted the role of lipid metabolism that confers the aggressive properties of malignant cancers. The current understanding of lipid metabolic reprogramming in cancer metastasis offers innovative and promising therapeutic implications.
Cancer cells fine-tune the anabolic/catabolic balance to meet increasing metabolic requirements and promote aggressive progression. During the progress of metastasis, cancer cells can couple lipid metabolism with supporting host cells in the microenvironment to benefit their distant metastasis.13
Crosstalk between glucose and lipid signals flows between metabolic and cell signaling networks that “acutely” regulate cell metabolism and proliferation, which, unfortunately, are “hijacked” by cancer cells. Cancer cells, if starved all glucose (ketogenesis) rewire metabolic programming and activate a aggressive ketogenesis pathway involving BRAF-MEK1 signaling.14
Cancer Progression is Not Simply Dependent on Glycolysis
Although the role of glycolysis has long been recognized in cancer progression (and is the reasoning behind the keto diet) the role of lipogenesis has recently been recognized as being equally or even more important.
In normal tissues, energy-providing lipids come principally from circulating lipids. However, in growing tumors, energy supply is mainly provided by lipids coming from de novo synthesis (the synthesis of complex molecules from simple molecules such as sugars or lipids, as opposed to recycling after partial degradation).
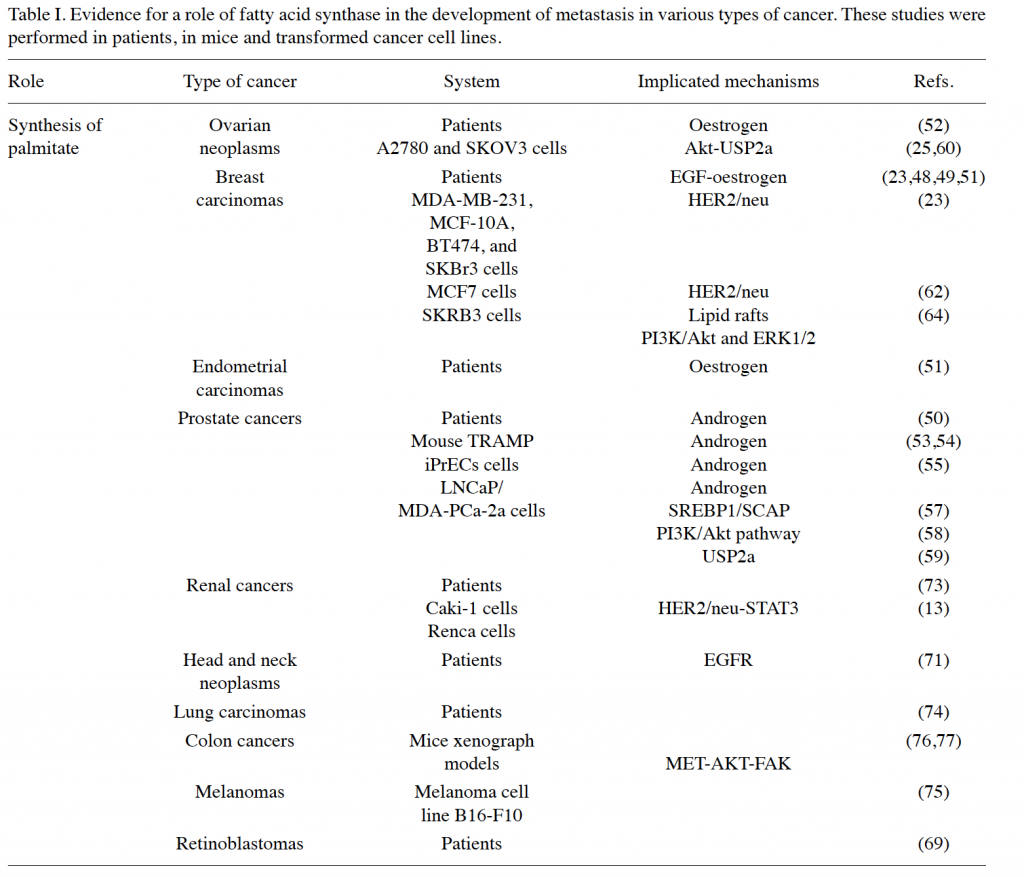
The role of lipogenic genes in the establishment of the primary tumor has been clearly established. A large number of studies demonstrate the role of fatty acid synthase in the activation of cell cycle and inhibition of apoptosis in tumor cells. Other lipogenic genes such as the acetyl CoA carboxylase and the stearoyl CoA desaturase 1 are highly expressed in primary tumors and also appear to play a role in their development.15
A Ketogenic Diet Can Actually Promote Tumor Growth
Ketogenic diets are sometimes used as anti-seizure therapy in patients with tuberous sclerosis, a rare genetic condition that causes mainly non-cancerous tumors throughout the body. Researchers have found that laboratory rats subjected to a ketogenic diet for 4, 6 and 8 months leads to excessive tumor growth, particularly with long-term usage.
The researchers noted that prolonged feeding of a ketogenic diet promoted the growth of renal tumors by recruiting ERK1/2 and mTOR, which are associated with the accumulation of oleic acid and the overproduction of growth hormone. Although there were initially some antitumor mechanisms triggered by the ketogenic diet, the pro-cancerous mechanisms took over and stimulated tumor growth. 16
Another study revealed a link between a ketogenesis pathway mechanism, finding that metabolic and cell signaling networks and signals are rewired to selectively promote tumor development. These discoveries represent the crosstalk of glucose and lipid signal flows with metabolic and cell signaling networks that regulate cell metabolism and proliferation, which, unfortunately, are hijacked by cancer cells.17
Although glycolysis is the best-known metabolic phenotype of cancers, it is not a unique feature of all human cancers. In fact, in highly glycolytic tumor cells, defects in mitochondrial oxidative metabolism are not found. Several studies indicate that under certain circumstances, cancer cells will depend on mitochondrial oxidative phosphorylation to meet with cellular ATP demands.18-19
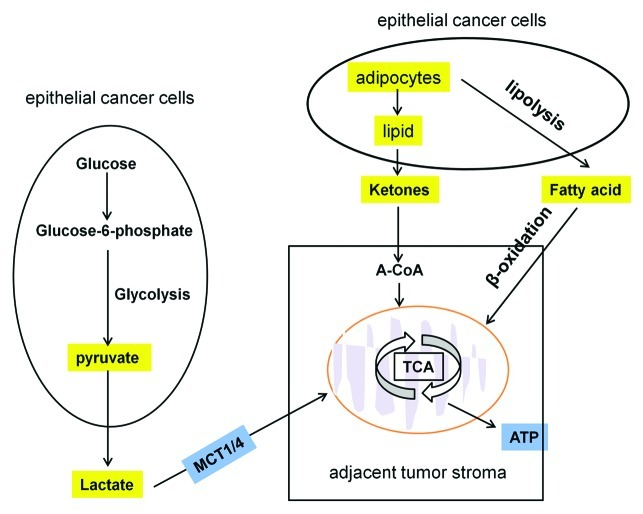
As evidenced by the illustration below, some energy-rich metabolites (L-lactate, ketones and fatty acids shown in yellow) derived from the tumor stroma can be transferred to adjacent cancer cells and used for energy production via mitochondrial oxidative phosphorylation.20
Effects of Ketogenic Diets on Cardiovascular Risk Factors
In addition to being promoted as a diet for cancer patients, the ketogenic diet is often promoted as a diet for preventing or treating cardiovascular disease. The results of ketogenic diets on cardiovascular risk factors are controversial, both in animals and humans, but some short-term improvements, notably in patients with obesity and type-2 diabetes, have been described.
Unfortunately, while the effects may initially prove beneficial, they don’t hold up over time, and can end up causing adverse effects. For example, in laboratory studies, ketogenic diets have been shown to cause nonalcoholic fatty liver disease (NAFLD) and insulin resistance.21
For more information, please refer to the previous blog posts I’ve written on this subject:
“Cancer: Why Quick Fix Solutions Fall Short—and Why ETMS Succeeds”
“Can A Ketogenic Diet Cure Cancer?”
In an upcoming post, I’ll discuss why I believe that the diet I recommend based on the principles of the Eclectic Triphasic Medical System (ETMS) is far superior to the ketogenic diet for improving and maintaining health.
Research
1. BradleyC. Johnston, PhD; Steve Kanters, MSc5,6,7; Kristofer Bandayrel, MPH1,4; et al Comparison of Weight Loss Among Named Diet Programs in Overweight and Obese Adults, A Meta-analysis, JAMA. 2014;312(9):923-933. doi:10.1001/jama.2014.10397
2. Li Ma, Yongzhen Tao, Angeles Duran, et al. Control of Nutrient Stress-Induced Metabolic Reprogramming by PKCz in Tumorigenesis. Cell, 2013; 152 (3): 599 DOI: 10.1016/j.cell.2012.12.028
3. Chung HY1, Park YK2. Rationale, Feasibility and Acceptability of Ketogenic Diet for Cancer Treatment. J Cancer Prev. 2017 Sep; 22(3): 127–134.
4. Jansen N, Walach H. The development of tumours under a ketogenic diet in association with the novel tumour marker TKTL1: A case series in general practice. Oncol Lett. 2016 Jan; 11(1):584-592. Epub 2015 Nov 16.
5. Rui Hai Liu, Potential Synergy of Phytochemicals in Cancer Prevention: Mechanism of Action, J. Nutr. December 1, 2004 vol. 134 no. 12 3479S-3485S
6. Zeng C, Toole BP, Kinney SD, Kuo JW, Stamenkovic I. Inhibition of tumor growth in vivo by hyaluronan oligomers, Int J Cancer. 1998 Jul 29;77(3):396-401.
7. Ghatak S, Misra S, Toole BP. Hyaluronan oligosaccharides inhibit anchorage-independent growth of tumor cells by suppressing the phosphoinositide 3-kinase/Akt cell survival pathway. J Biol Chem. 2002 Oct 11;277(41):38013-20. Epub 2002 Jul 26.
8. Ward JA, Huang L, Guo H, Ghatak S, Toole BP. Perturbation of hyaluronan interactions inhibits malignant properties of glioma cells.Ward JA, Huang L, Guo H, Ghatak S, Toole BP. Am J Pathol. 2003 May;162(5):1403-9.
9. Choi SP, Kim SP, Nam SH, Friedman M. Antitumor effects of dietary black and brown rice brans in tumor-bearing mice: relationship to composition. Mol Nutr Food Res. 2013 Mar;57(3):390-400. doi: 10.1002/mnfr.201200515. Epub 2012 Dec 23.
10. Shakib MC , Gabrial SG 1 , Gabrial GN 1 Beetroot-Carrot Juice Intake either Alone or in Combination with Antileukemic Drug ‘Chlorambucil’ As A Potential Treatment for Chronic Lymphocytic Leukemia. Open Access Maced J Med Sci. 2015 Jun 15;3(2):3316. doi: 10.3889/oamjms.2015.056. Epub 2015 Jun 2.
11. Zhang B, Zhao Q, Guo W, Bao W, Wang X. Eur J Clin Nutr. Association of whole grain intake with all-cause, cardiovascular, and cancer mortality: a systematic review and dose-response meta-analysis from prospective cohort studies 2017 Nov 1. doi: 10.1038/ejcn.2017.149.
12. Ubaldo E. Martinez-Outschoorn et. al., Ketones and lactate increase cancer cell “stemness”, driving recurrence, metastasis and poor clinical outcome in breast cancer, Cell Cycle 10:8, 1271-1286; April 15, 2011; © 2011 Landes Bioscience
13. Xiangjian Luo, Can Cheng, Zheqiong Tan, Namei Li, Min Tang, Lifang Yang, and Ya Cao, Emerging roles of lipid metabolism in cancer metastasis, Mol Cancer. 2017; 16: 76, Published online 2017 Apr 11. doi: 10.1186/s12943-017-0646-3.
14. Hee-Bum Kang et. al., Metabolic rewiring by oncogenic BRAF V600E links ketogenesis pathway to BRAF-MEK1 signaling, Mol Cell. 2015 August 6; 59(3): 345–358. doi:10.1016/j.molcel.2015.05.037.
15. Mounier C, Bouraoui L, and Rassart E. Lipogenesis in cancer progression, International Journal of Oncology 45: 485-492, 2014.
16. Liśkiewicz AD, Kasprowska D, Wojakowska A, Polański K, Lewin-Kowalik J, Kotulska K, Jędrzejowska-Szypułka H. Longterm High Fat Ketogenic Diet Promotes Renal Tumor Growth in a Rat Model of Tuberous Sclerosis, Sci Rep. 2016 Feb 19;6:21807. doi: 10.1038/srep21807.
17. Hee-Bum Kang et. al., Metabolic rewiring by oncogenic BRAF V600E links ketogenesis pathway to BRAF-MEK1 signaling, Mol Cell. 2015 August 6; 59(3): 345–358. doi:10.1016/j.molcel.2015.05.037.
18. Goldberg MS, Sharp PA. Pyruvate kinase M2-specific siRNA induces apoptosis and tumor regression. J Exp Med. 2012;209:217–24. doi: 10.1084/jem.20111487.
19. Walenta S, Wetterling M, Lehrke M, Schwickert G, Sundfør K, Rofstad EK, et al. High lactate levels predict likelihood of metastases, tumor recurrence and restricted patient survival in human cervical cancers. Cancer Res. 2000;60:916–21.
20. Yi Zhang 1 and Jin-Ming Yang. Altered energy metabolism in cancer, Cancer Biol Ther. Feb 1, 2013; 14(2): 81–89.
21. Kosinski C and Jornayvaz F. Effects of Ketogenic Diets on Cardiovascular Risk Factors: Evidence from Animal and Human Studies, Nutrients 2017, 9, 517; doi:10.3390/nu9050517.


Wow! I thoroughly enjoyed reading this article! It well explained, and full of common sense! Thank you so much for your effort! I’ve discovered your site recently, but I can’t stop reading it! Best wishes!