My intention for this article is to give you scientifically valid information about the Covid-19 vaccines so that you can make an educated decision about whether or not to get one. Personally, I have complete faith in the tools I employ to optimize my own health and support my immune system to protect me from becoming ill. My opinion is that for those who are not in great health or are not taking a health supportive protocol (including supplementing with, at a minimum, vitamin D and zinc), those over age seventy-five, anyone who is obese, and those with a pre-existing condition(s), the vaccine would be a wise choice given the high risk of long-term complications and death in these populations.
In addition to all the many benefits of herbal medicines against viral infections, which I will discuss here further, nutrition is equally as important and essential to boosting the immune system. According to the World Health Organization, healthy foods rich in vitamins and minerals and adequate hydration are vital to wellness. Individuals consuming a well-balanced diet are healthier with a strong immune system and have a reduced risk of chronic illness and infectious diseases. Insufficient levels of zinc, vitamin D, selenium, vitamin C, vitamin E, and other important nutrients can lead to coronavirus infection.[1],[2],[3] Zinc in particular plays a critical role in all components of the immune system. A brand-new study just published found that low zinc levels are associated with severity of Acute Respiratory Distress Syndrome by Covid-19. [4] Eating a diverse, wholesome, plant-based diet and supplementing with these key nutrients has never been more important!
Immunity at a Glance
The body has two primary types of immunity: humoral and cell mediated. Humoral immunity involves the rapid or quick response against antigens, while cell mediated immunity works in a slower but more permanent fashion against antigens. In humoral immunity responses, B cells produce antibodies after being activated by free antigens present in body fluids. In cell mediated immunity responses, T cells attack infected body cells that display the antigens of pathogens on their surface.
Traditional Vaccine Technologies vs. Covid-19 Vaccine Technology
Traditionally, vaccines have been created either by using an inactivated form of the virus or by attenuating the virus to make it less infectious. Proteins from the modified viruses stimulate the production of B and T cells without triggering symptoms of a real infection. The body keeps a few T cells, also called memory cells, that go into action quickly if the body encounters the same virus again. Additionally, when the familiar antigens are detected, B cells quickly produce antibodies to attack them.[5] The idea is to bypass (trick) the cell mediated immune system to go directly to antibody production, thereby creating cells that recognize the virus and can eliminate it on contact. I have written about traditional vaccines and the immune response in past blogs, so you can read them if you would like to know more.
The first and second Covid-19 vaccines in the U.S. have now been granted emergency use authorization and are initially being distributed to frontline healthcare providers and assisted living residents. These new vaccines, produced by Moderna and Pfizer, employ an mRNA technology that is the first of its kind and has never been approved or authorized for human use before, but has been studied. In my opinion, there is a great deal more to learn about the potential long-term effects the new vaccines may have in order to ensure their safety. For those concerned about excipients used in licensed vaccines, a useful resource can be downloaded here (does not include the new Covid-19 vaccine excipients).
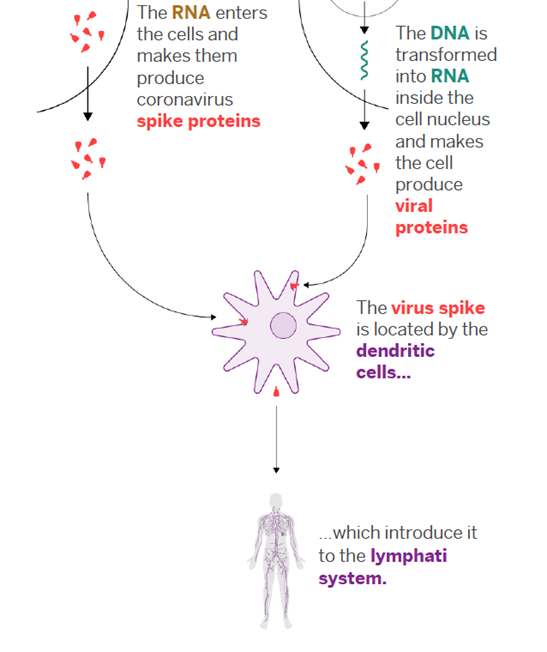
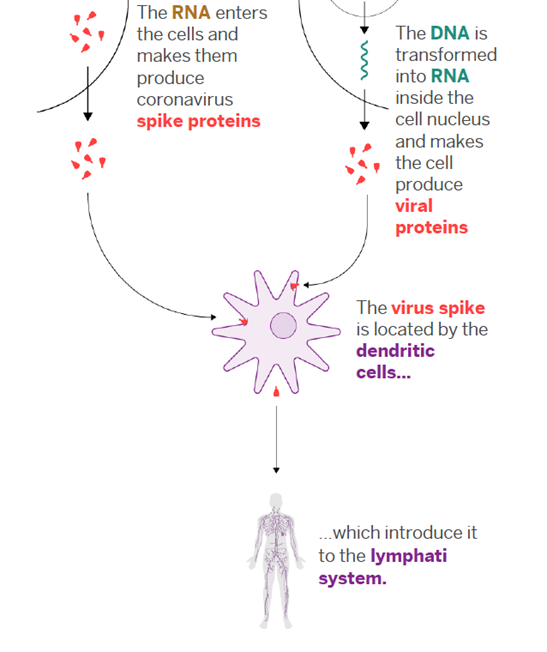
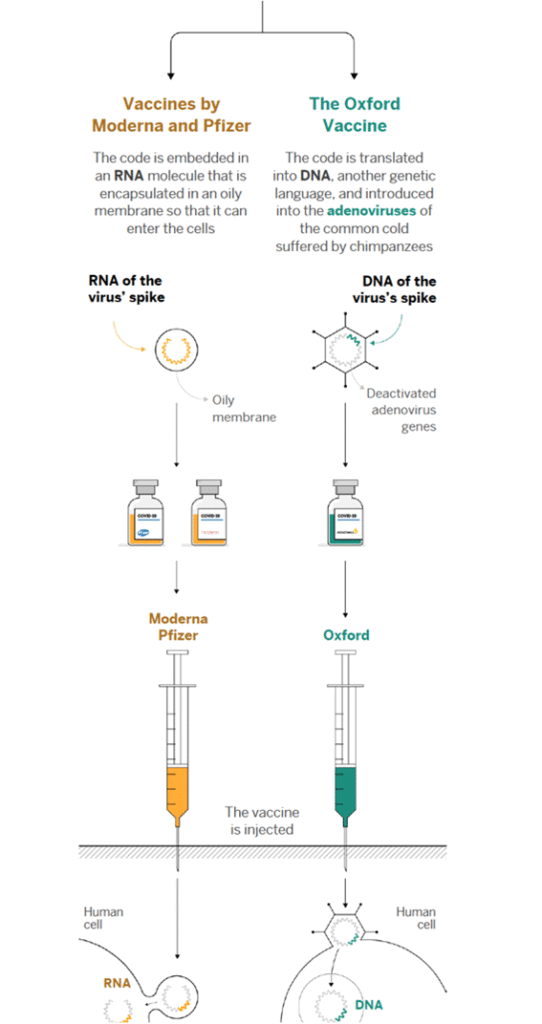
In this new technology, the viral mRNA sequence that codes for the required viral protein is injected into the body, versus a modified version of the virus itself. The body itself translates this injected viral mRNA sequence into the viral protein that is required for the proper immune response. In the case of Covid-19, we’re interested in the “spike protein” on the surface of the virus. [6]
The price of each dose is an essential factor in making a drug accessible to all. The more traditional vaccine developed by Oxford University and the biopharmaceutical company AstraZeneca, will cost around $3 per dose, while the one developed by the US multi-national Pfizer and the German biotech company BioNTech will cost more than $15 a dose, and the one developed by the US company Moderna and the US National Institutes of Health will cost $21 a dose.
One big advantage of an mRNA vaccine is that it is very easy to produce on a large scale. Because it is completely synthetic, it does not have to be grown in cells, unlike the traditionally produced vaccines. So, it is easier to make and the costs are much lower, although the price to the consumer will be much higher.
Weighing the Pros and Cons of Vaccine Technologies
The technology employed by both vaccines to inject the material into the cells is called transfection, a process by which ribonucleic acids or nucleic acids (RNA or DNA) are introduced into cells, utilizing means other than viral vectors (carriers). The vaccines employ a “hydrogel nanotechnology” which refers to the lipid nanoparticles (small oil droplets) that encase the mRNA. Because our cell membranes are composed of lipids, when the vaccine is injected, the vaccine lipid particles merge with the lipid on the surface of the cell (like two oil droplets merging) and the piece of mRNA is delivered inside the cell. [7]
Oxford University has stated that its vaccine can be kept in a refrigerator, at temperatures of between 2° and 8° Celsius, and can therefore be distributed using the existing channels for other vaccines. Meanwhile, Pfizer’s vaccine requires ultra-cold temperatures of about -70° Celsius, a problem the company will try to solve by using dry ice containers that can keep the vaccine at the right temperature for 15 days. The storage temperature for the Moderna vaccine is somewhere between the two. [8]
Vaccine Effectiveness
The Coronavirus Efficacy (COVE) phase 3 trial was launched in late July 2020 to assess the safety and efficacy of the Moderna COVID-19 (mRNA-1273) vaccine in preventing SARS-CoV-2 infection. According to this study just published, the mRNA-1273 vaccine showed 94.1% efficacy at preventing Covid-19 illness, including severe disease. Aside from transient local and systemic reactions, no safety concerns were identified.[9] The anecdotal finding of a slight excess of Bell’s palsy in this trial and in the Pfizer-BioNTech (BNT162b2) vaccine trial arouses concern that it may be more than a chance event, and the possibility bears close monitoring.[10]
Researchers at MIT’s Computer Science and Artificial Intelligence Lab (CSAIL) showed that the vaccines’ effectiveness may vary depending on a person’s race and said that the vaccines should be tested robustly across populations with diverse genetic backgrounds. Using advanced machine learning AI methods that examined a form of vaccine similar to Moderna and Pfizer’s, the team found that the number of people whose cellular immune system is not predicted to robustly respond to the vaccine ranged from less than half of one percent of white participants to nearly 10 percent of Asian participants (participants self-reported their race.)[11]
Additionally, the effectiveness of the vaccine may also vary according to age. For example, in young, healthy adults, it appears that psychological stress is detrimental to long-term maintenance of antibody levels following vaccination.[12] In another study, elderly caretakers of spouses with dementia were found to have increased activation of the hypothalamic-pituitary-adrenal (HPA) axis and a poor antibody response to influenza vaccine. [13] This suggests that the most vulnerable population, the elderly, may require more comprehensive support in order to be fully protected from the risk of Covid-19 even with the vaccine, given the possibility of it being less effective in this population. Certainly a role for adaptogenic herbs, immune tonics, vitamin D and zinc taken with the Covid-19 vaccine should be explored, particularly in the elderly.
Research on Traditional Herbal Medicines for Combatting SARS-CoV-2
While researchers around the globe race towards discovering effective treatments or cures for Covid-19, traditional herbal medicine has been largely ignored despite the on-going positive results demonstrated with its use. Nature provides a huge reservoir of anti-infectious compounds and while there have been no absolute therapies for Covid‐19 until recently, herbal and natural remedies can be used to support the body’s natural response to infection and to control further complications and organ damage.[14],[15]
The herbal remedy forsythia suspensa, which has been traditionally used for hundreds of years to combat flu viruses, was put to the test for SARS virus in laboratory studies and was observed to have quite positive results. An ingredient of forsythia suspensa, called Phillyrin (KD-1), possesses anti-inflammatory, anti-oxidant, and anti-viral activities, demonstrating it could significantly inhibit SARS-CoV-2 and HCoV-229E replication in vitro. The study suggests KD-1 could also markedly reduce the production of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β, MCP-1, and IP-10) at the mRNA levels.[16]
A traditional Chinese medicine (TCM) formula that features Forsythia suspensa and Lonicera japonica, was found to exert broad-spectrum anti-viral effects on a series of influenza viruses and recently, to significantly inhibit the SARS-COV-2 replication, affect virus morphology, and exert anti-inflammatory activity in vitro.[17] In the following multicenter, prospective, randomized controlled trial, patients were found to have a greater recover rate, shorter recovery time and less physical damage from Covid-19.
“The study was divided into two arms. One group received usual care and another received usual care plus LH Capsules (4 pills, 3X daily) for a total of 14 days. Out of a total of 284 patients in the study (142 in each group), the group receiving LH capsules had a greater recovery rate (91.5%) than the group receiving only usual care (82.4%).
The median time to symptomatic recovery was 7 days in the LH Capsules group and 10 days in the usual care monotherapy group. Symptom recovery times in the LH capsules group were significantly better than the usual care only group. The LH capsules group had the following symptom recovery rates: 57.7% day 5, 80.3% day 10, 91.5% day 14.
Similar results were found across several other parameters. The LH Capsules group had a shorter recovery time from “fever (2 vs. 3 days), fatigue (3 vs. 6 days) and coughing (7 vs. 10 days).” Importantly, chest computed tomographic (CT) improvements were significantly greater in the LH Capsules group (83.8% vs. 64.1%). This indicates less physical damage to patients from COVID-19 (coronavirus) for patients receiving LH Capsules. In addition, the total clinical recovery rate was better in the LH Capsules group (78.9% vs. 66.2%).”[18]
Additionally, during the outbreak of SARS in China, 50 ± 10 % infected patients were given the traditional Chinese medicine (TCM) along with modern medical procedures, at some stage of their therapy. The results reveal positive, but inconclusive outcomes regarding the efficacy of the combined treatments using Chinese medicine as an adjuvant. Positive effects using adjuvant herbal therapy included better control of fever, quicker clearance of chest infection, lesser consumption of steroids and other symptoms were relieved. In a few reports, some evidence of immunological bolstering was also found. This synergistic administration was approved and supported by the Chinese Center for Disease Control and Prevention, and further information on the herbal treatment were recommended to other practitioners.[19]
Soshihotang (SSHT, Xiao Chai Hu Tang in Chinese) has been prescribed to treat various viral diseases and is used in combination with other herbal medicines depending on the patient’s symptoms. SSHT is made up of Bupleurium, Scutellaria b., Pinellia, Ginseng Radix, Zingiber, Ziziphus, and glycyrrhiza. In a number of recent Covid-19 clinical studies, treatment with SSHT improved the infection status of the respiratory and hepatobiliary systems, and experimental studies demonstrated the antiviral effect of SSHT and its components. [20]
Furthermore, according to the Indian government, traditional Siddha and Ayurveda treatments have completely cured 30 COVID-19 patients.[21] Ashwagandha, a well-known Ayurvedic medicine and primary adaptogen, demonstrated inhibition against a main protease of SARS-CoV-2.[22]
Due to the wide variability in chemical superstructure of the components of assorted herbal extracts, common motifs have been identified that demonstrate effective anti-viral activity against SARS-CoV-2. Looking at the most active compounds in each extract, it becomes evident that as a group, phenolic compounds have a broad enzyme inhibiting function.[23]
Echinacea as an Important Ally to Immune Health

Echinacea species have traditionally been used in North America for the prevention and treatment of infections, including the common cold and flu. Various animal studies suggest that Echinacea may modulate innate and adaptive immune responses, as well as antibody production.[24] Echinacea extracts have also been used for the prevention and treatment of respiratory tract infections, with beneficial effects documented in both preclinical and clinical studies. When assessing all human trials (17 studies in total, including 3363 participants) which reported changes in cytokine levels in response to Echinacea supplementation, the results were largely consistent with a decrease in the pro-inflammatory cytokines that play a role in the progression of cytokine storm and Acute Respiratory Distress Syndrome (ARDS), factors that play a significant role in the death of COVID-19 patients.[25] Recently, Echinacea extract (Echinaforce®) was shown to be virucidal against HCoV-229E and MERS-CoV and both SARS-CoV-1 and SARS-CoV-2 were inactivated at similar concentrations of the extract.[26]
Echinacea purpurea extract, along with zinc, selenium and vitamin C, alleviated exacerbations of chronic obstructive pulmonary disease according to results from a randomized controlled trial.[27]
Also, according to a recent 3-week human study, echinacea (Echinacea angustifolia root) extract was shown to decrease anxiety in subjects,[28] which may provide further benefit since stress weakens the immune and nervous systems.
I often combine echinacea with several other herbs, including propolis. Propolis has been proven to possess anti-viral activity both in vitro and in vivo. In pre-clinical studies, propolis promoted immunoregulation of pro-inflammatory cytokines, including reduction in IL-6, IL-1 beta and TNF-α. This immunoregulation involves monocytes and macrophages, as well as Jak2/STAT3, NF-kB, and inflammasome pathways, which reduce the risk of cytokine storm syndrome, a major mortality factor in advanced COVID-19 disease.[29]
Additionally, Echinacea purpurea extract has shown to improve antibody production when taken with an influenza vaccine. In one study, Echinacea increased (P<0.05) the antibody titer in comparison to controls. A dose of 0.5% had a better effect than 0.1% in an ELISA test. Thus, Echinacea had an enhancing effect on the efficiency of an influenza vaccine.[30]
The Role of Adaptogens in Immune Support
Adaptogenic herbs play a key role in regulating various metabolic processes through improvement in HPA functioning and stress response. Adaptogens have been traditionally used as tonics to help build strength after illness and trauma. They have also been demonstrated time and time again to support immune function and reduce susceptibility to illness.
Some Major Adaptogenic Herbs Include the Following:
Astragalus membranaceus (Astragalus): Astragalus is primarily used as a lung tonic and may be helpful in increasing resistance against respiratory infections. Traditionally, Astragalus has been used to invigorate Qi.[32] It has been shown to potentiate both thymus and spleen function and to augment both humoral and cell-mediated immunity.[33]
Eleuthero (Eleutherococcus senticosus ): Eleuthero (formerly known as Siberian Ginseng) has primarily been valued for its ability to increase nonspecific resistance to physical and psychological stresses. It stimulates T-lymphocyte function and increases macrophage activity in lymph tissue.[34]
Reishi (Ganoderma lucidum): Reishi is one of the primary herbs of choice in any immune deficiency disease because it possesses a broad spectrum of immune stimulating activities, as well as anti-inflammatory and antiallergenic properties. Reishi contains more than 100 oxygenated triterpenes, many of which exhibit a marked effect on the activity of NK cells. Reishi has been widely used for a variety of infectious diseases, such as bronchitis and hepatitis. It stimulates phagocytosis, increases T-cell activity and is a treatment for viral hepatitis. Reishi has been reported to increase CD4 cells in vivo.[35],[36],[37],[38],[39],[40],[41],[42]
Ginseng (Panax ginseng): Ginseng is tonifying, increases stamina, spares muscle use of carbohydrates and stimulates hypothalamic output and ACTH and therefore adrenal cortex function. Ginseng is used primarily to improve psychological function, physical vitality, and activity. It is further known to support immune function through aiding convalescence and as a prophylactic to build resistance and reduce susceptibility to illness.[43]
The aforementioned species are just a few important traditional herbs that could be of benefit. I highly recommend that people over the age of 50 supplement with adaptogenic and tonifying herbs to help counteract age-related changes at the epigenomic, cellular and organ system levels. I prefer to use combination formulas over individual herbs, however. For an in-depth review of these and other important adaptogenic herbs, I suggest you read my book entitled, “Adaptogens in Medical Herbalism.” For additional specific recommendations on how to optimize your health if you do decide to get the vaccine, please contact the Mederi Center for a consult with one of our practitioners.
[1] C.J. Field, I.R. Johnson, P.D. Schley, Nutrients and their role in host resistance to infectionJ Leukoc Biol, 71 (2002), pp. 16-32
[2] M. Maares, H. Haase, Zinc and immunity: an essential interrelation, Arch Biochem Biophys, 611 (2016), pp. 58-65
[3] T.B. Ng, R.C. Cheung, J.H. Wong, Y. Wang, D.T. Ip, D.C. Wan, et al. Antiviral activities of whey proteins, Appl Microbiol Biotechnol, 99 (2015), pp. 6997-7008
[4] Gonçalves TJM, Gonçalves SEAB, Guarnieri A, Risegato RC, Guimarães MP, de Freitas DC, Razuk-Filho A, Junior PBB, Parrillo EF. Association Between Low Zinc Levels and Severity of Acute Respiratory Distress Syndrome by New Coronavirus (SARS-CoV-2). Nutr Clin Pract. 2020 Dec 23. doi: 10.1002/ncp.10612. Epub ahead of print. PMID: 33368619.
[5] CDC website, National Center for Immunization and Respiratory Diseases (NCIRD), Division of Viral Diseases, Understanding How COVID-19 Vaccines Work, 11/02/2020 https://www.cdc.gov/coronavirus/2019-ncov/vaccines/different-vaccines/how-they-work.html, retrieved 12/10/2020
[6] Peters, Adele is a staff writer at Fast Company, How Pfizer and Moderna’s COVID-19 vaccines work: mRNA, 11/09/20, https://www.fastcompany.com/90573488/how-pfizers-covid-19-vaccine-works-mrna, retrieved 11/18/20.
[7] Marshall, Serena, Salahi, Lara, December 9, 2020, MEDPAGE TODAY, How Do the New COVID-19 Vaccines Work? Richard Kuhn, PhD, discusses the technology behind the new mRNA vaccines, https://www.medpagetoday.com/podcasts/trackthevax/90085?xid=nl_mpt_investigative2020-12-09&eun=g1065123d0r&utm_source=Sailthru&utm_medium=… , retrieved 12/9/2020
[8] MANUEL ANSEDE | ARTUR GALOCHA , The differences between the Pfizer, Moderna and Oxford coronavirus vaccines: what we know so far, Science and Tech, Dec, 2020
[9] Haynes Barton F. et.al., (2020) A New Vaccine to Battle Covid-19. N Engl J Med DOI: 10.1056/NEJMe2035557. Funded by the Biomedical Advanced Research and Development Authority and the National Institute of Allergy and Infectious Diseases; COVE ClinicalTrials.gov number, NCT04470427. opens in new tab.
[10] Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT162b2 Covid-19 vaccine. N Engl J Med 2020;383:2603-2615.
[11] Liu, Ge et al. Predicted Cellular Immunity Population Coverage Gaps for SARS-CoV-2 Subunit Vaccines and their Augmentation by Compact Peptide Sets, Cell Systems, Published: November 26, 2020DOI:https://doi.org/10.1016/j.cels.2020.11.010.
[12] Burns VE, Carroll D, Drayson M, Whitham M, Ring C. Life events, perceived stress and antibody response to influenza vaccination in young, healthy adults. J Psychosom Res. 2003 Dec;55(6):569-72. doi: 10.1016/s0022-3999(03)00073-4. PMID: 14642989.
[13] Vedhara K, Cox NK, Wilcock GK, Perks P, Hunt M, Anderson S, Lightman SL, Shanks NM. Chronic stress in elderly carers of dementia patients and antibody response to influenza vaccination. Lancet. 1999 Feb 20;353(9153):627-31. doi: 10.1016/S0140-6736(98)06098-X. PMID: 10030328.
[14] Adhikari B, Marasini BP, Rayamajhee B, et al. Potential roles of medicinal plants for the treatment of viral diseases focusing on COVID-19: A review [published online ahead of print, 2020 Oct 9]. Phytother Res. 2020;10.1002/ptr.6893. doi:10.1002/ptr.6893
[15] Hensel A, Bauer R, Heinrich M, Spiegler V, Kayser O, Hempel G, Kraft K. Challenges at the Time of COVID-19: Opportunities and Innovations in Antivirals from Nature. Planta Med. 2020 Jul;86(10):659-664. doi: 10.1055/a-1177-4396. Epub 2020 May 20. PMID: 32434254; PMCID: PMC7356065.
[16] Ma Q, Li R, Pan W, Huang W, Liu B, Xie Y, Wang Z, Li C, Jiang H, Huang J, Shi Y, Dai J, Zheng K, Li X, Hui M, Fu L, Yang Z. Phillyrin (KD-1) exerts anti-viral and anti-inflammatory activities against novel coronavirus (SARS-CoV-2) and human coronavirus 229E (HCoV-229E) by suppressing the nuclear factor kappa B (NF-κB) signaling pathway. Phytomedicine. 2020 Nov;78:153296. doi: 10.1016/j.phymed.2020.153296. Epub 2020 Aug 1. PMID: 32890913; PMCID: PMC7395229.
[17] Runfeng L, Yunlong H, Jicheng H, Weiqi P, Qinhai M, Yongxia S, Chufang L, Jin Z, Zhenhua J, Haiming J, Kui Z, Shuxiang H, Jun D, Xiaobo L, Xiaotao H, Lin W, Nanshan Z, Zifeng Y. Lianhuaqingwen exerts anti-viral and anti-inflammatory activity against novel coronavirus (SARS-CoV-2). Pharmacol Res. 2020 Jun;156:104761. doi: 10.1016/j.phrs.2020.104761. Epub 2020 Mar 20. PMID: 32205232; PMCID: PMC7102548.
[18] Hu, Ke, Wei-jie Guan, Ying Bi, Wei Zhang, Lanjuan Li, Boli Zhang, Qingquan Liu et al. “Efficacy and Safety of Lianhuaqingwen Capsules, a repurposed Chinese Herb, in Patients with Coronavirus disease 2019: A multicenter, prospective, randomized controlled trial.” Phytomedicine (2020): 153242.
[19] Leung PC. The efficacy of Chinese medicine for SARS: A review of Chinese publications after the crisis. Am. J. Chin. Med., 2007; 35(4): 575-581. doi:10.1142/S0192415X07005077.
[20] Kwon S, Lee W, Jin C, et al. Could herbal medicine (Soshihotang) be a new treatment option for COVID-19?: a narrative review. Integr Med Res. 2020;9(3):100480. doi:10.1016/j.imr.2020.100480
[21] http://newsonair.com/News?title=Traditional-Indian-system-of-medicine-Siddha-proves-to-be-effective-in-Covid-19-management&id=391212
[22] Tripathi MK, Singh P, Sharma S, Singh TP, Ethayathulla AS, Kaur P. Identification of bioactive molecule from Withania somnifera (Ashwagandha) as SARS-CoV-2 main protease inhibitor [published online ahead of print, 2020 Jul 8]. J Biomol Struct Dyn. 2020;1-14. doi:10.1080/07391102.2020.1790425
[23] Wyganowska-Swiatkowska M, Nohawica M, Grocholewicz K, Nowak G. Influence of Herbal Medicines on HMGB1 Release, SARS-CoV-2 Viral Attachment, Acute Respiratory Failure, and Sepsis. A Literature Review. Int J Mol Sci. 2020 Jun 30;21(13):4639. doi: 10.3390/ijms21134639. PMID: 32629817; PMCID: PMC7370028.
[24] Bałan, B. J., Sokolnicka, I., Skopińska-Różewska, E., & Skopiński, P. (2016). The modulatory influence of some Echinacea-based remedies on antibody production and cellular immunity in mice. Central European Journal of Immunology, 1(1), 12–18. doi: 10.5114/ceji.2016.58813
[25] Aucoin M, Cooley K, Saunders PR, Carè J, Anheyer D, Medina DN, Cardozo V, Remy D, Hannan N, Garber A. The effect of Echinacea spp. on the prevention or treatment of COVID-19 and other respiratory tract infections in humans: A rapid review. Adv Integr Med. 2020 Dec;7(4):203-217. doi: 10.1016/j.aimed.2020.07.004. Epub 2020 Aug 1. PMID: 32837894; PMCID: PMC7395221.
[26] Signer J, Jonsdottir HR, Albrich WC, Strasser M, Züst R, Ryter S, Ackermann-Gäumann R, Lenz N, Siegrist D, Suter A, Schoop R, Engler OB. In vitro virucidal activity of Echinaforce®, an Echinacea purpurea preparation, against coronaviruses, including common cold coronavirus 229E and SARS-CoV-2. Virol J. 2020 Sep 9;17(1):136. doi: 10.1186/s12985-020-01401-2. Erratum in: Virol J. 2020 Nov 9;17(1):172. PMID: 32907596; PMCID: PMC7479405.
[27] Isbaniah F, Wiyono WH, Yunus F, Setiawati A, Totzke U, Verbruggen MA. Echinacea purpurea along with zinc, selenium and vitamin C to alleviate exacerbations of chronic obstructive pulmonary disease: results from a randomized controlled trial. J Clin Pharm Ther. 2011 Oct;36(5):568-76. doi: 10.1111/j.1365-2710.2010.01212.x. Epub 2010 Nov 10. PMID: 21062330.
[28] Haller J, Krecsak L, Zámbori J. Double-blind placebo controlled trial of the anxiolytic effects of a standardized Echinacea extract. Phytother Res. 2020;34(3):660-668. doi:10.1002/ptr.6558
[29] Berretta AA, Silveira MAD, Cóndor Capcha JM, De Jong D. Propolis and its potential against SARS-CoV-2 infection mechanisms and COVID-19 disease: Running title: Propolis against SARS-CoV-2 infection and COVID-19 [published online ahead of print, 2020 Aug 17]. Biomed Pharmacother. 2020;131:110622. doi:10.1016/j.biopha.2020.110622
[30] Najafzadeh H., et. al., Effect of Echinacea purpurea on antibody production against fowl influenza vaccine, June 2011, Journal of Applied Animal Research 39(2):139-141
[31] Ann. N.Y. Acad. Sci. 1401 (2017) 49–64 C 2017 Panossian, A., G. Wikman & H. Wagner. 1999. Plant adaptogens. III. Earlier and more recent aspects and concepts on their mode of action. Phytomedicine 6: 287–300.
[32] Bian YQ, Li J, Peng S, Lyu TY, Zhang YL, Qiao YJ. [Exploration of potential efficacy markers of Astragali Radix for invigorating Qi based on systematic traditional Chinese medicine]. Zhongguo Zhong Yao Za Zhi. 2020 Jul;45(14):3266-3274. Chinese. doi: 10.19540/j.cnki.cjcmm.20200210.405. PMID: 32726039.
[33] Guo Z, Lou Y, Kong M, Luo Q, Liu Z, Wu J. A Systematic Review of Phytochemistry, Pharmacology and Pharmacokinetics on Astragali Radix: Implications for Astragali Radix as a Personalized Medicine. Int J Mol Sci. 2019 Mar 22;20(6):1463. doi: 10.3390/ijms20061463. PMID: 30909474; PMCID: PMC6470777.
[34] Bone, K. Mills, S. Herbal approaches to pathological states In Principles and Practice of Phytotherapy (Second Edition), 2013; https://www.sciencedirect.com/topics/medicine-and-dentistry/adaptogenic, retrieved 12/20/2020
[35] Wang X, Lin Z. Immunomodulating Effect of Ganoderma (Lingzhi) and Possible Mechanism. Adv Exp Med Biol. 2019;1182:1-37. doi: 10.1007/978-981-32-9421-9_1. PMID: 31777013.
[36] Chen L, Abulizi A, Li M. Protective Effect of Ganoderma (Lingzhi) on Radiation and Chemotherapy. Adv Exp Med Biol. 2019;1182:119-142. doi: 10.1007/978-981-32-9421-9_4. PMID: 31777016.
[37] Wang C, Shi S, Chen Q, Lin S, Wang R, Wang S, Chen C. Antitumor and Immunomodulatory Activities of Ganoderma lucidum Polysaccharides in Glioma-Bearing Rats. Integr Cancer Ther. 2018 Sep;17(3):674-683. doi: 10.1177/1534735418762537. Epub 2018 Apr 2.
[38] Chang YH, Yang JS, Yang JL, et al. Ganoderma lucidum extracts inhibited leukemia WEHI-3 cells in BALB/c mice and promoted an immune response in vivo. Biosci Biotechnol Biochem. 2009 Dec;73(12):2589-94.
[39] Boh B, Berovic M, Zhang J, Zhi-Bin L. Ganoderma lucidum and its pharmaceutically active compounds. Biotechnol Annu Rev. 2007;13:265-301
[40] Wachtel-Galor S, Yuen J, Buswell JA, Benzie IFF. Ganoderma lucidum (Lingzhi or Reishi): A Medicinal Mushroom. In: Benzie IF, Wachtel-Galor S, eds. Herbal Medicine: Biomolecular and Clinical Aspects. 2nd ed. Boca Raton: CRC Press; 2011.
[41] Shi Y, Sun J, He H, Guo H, Zhang S. Hepatoprotective effects of Ganoderma lucidum peptides against D-galactosamine-induced liver injury in mice. J Ethnopharmacol. 2008 May 22;117(3):415-9.
[42] Dudhgaonkar S, Thyagarajan A, Sliva D. Suppression of the inflammatory response by triterpenes isolated from the mushroom Ganoderma lucidum. Int Immunopharmacol. 2009 Oct;9(11):1272-80.
[43] Maggini, S., Wishart, E., Wintergerst, S., Foods and Dietary Supplements in the Prevention and Treatment of Disease in Older Adults, 2015, Chapter 25 – Micronutrients and Ginseng for Immune Support in Older Adults, Pages 265-275, https://doi.org/10.1016/B978-0-12-418680-4.00025-7